I am opening this thread to share some information on vitamin B12 deficiency which can go unnoticed or misdiagnosed because of inferior testing methods. It is a pattern that I have noticed in many individuals who present with neurological symptoms and chronic fatigue/fibromyalgia type symptoms, yet have normal or high serum vitamin B12 on an ordinary blood panel.
This may lead a clinician to assume that B12 levels are optimal. However, the high serum B12 can actually be indicative of a functional problem with transporting it into the cells and then utilizing it properly. Hence, the B12 deficiency is masked by the "unremarkable" blood testing results. This is the main reason why I find urinary/blood methylmalonic acid as the best marker for B12 deficiency, along with homocysteine as a supporting marker. I have consulted with numerous people who had high serum B12, but markers of severe functional deficiency. So maybe there will be some people on the forum who might find this information helpful.
This may lead a clinician to assume that B12 levels are optimal. However, the high serum B12 can actually be indicative of a functional problem with transporting it into the cells and then utilizing it properly. Hence, the B12 deficiency is masked by the "unremarkable" blood testing results. This is the main reason why I find urinary/blood methylmalonic acid as the best marker for B12 deficiency, along with homocysteine as a supporting marker. I have consulted with numerous people who had high serum B12, but markers of severe functional deficiency. So maybe there will be some people on the forum who might find this information helpful.
Biomarkers and Algorithms for the Diagnosis of Vitamin B12 Deficiency
Vitamin B12 (cobalamin, Cbl, B12) is an indispensable water-soluble micronutrient that serves as a coenzyme for cytosolic methionine synthase (MS) and mitochondrial methylmalonyl-CoA mutase (MCM). Deficiency of Cbl, whether nutritional or due to inborn errors of Cbl metabolism, inactivate MS and MCM leading to the accumulation of homocysteine (Hcy) and methylmalonic acid (MMA), respectively. In conjunction with total B12 and its bioactive protein-bound form, holo-transcobalamin (holo-TC), Hcy, and MMA are the preferred serum biomarkers utilized to determine B12 status. Clinically, vitamin B12 deficiency leads to neurological deterioration and megaloblastic anemia, and, if left untreated, to death. Subclinical vitamin B12 deficiency (usually defined as a total serum B12 of <200 pmol/L) presents asymptomatically or with rather subtle generic symptoms that oftentimes are mistakenly ascribed to unrelated disorders. Numerous studies have now established that serum vitamin B12 has limited diagnostic value as a stand-alone marker. Low serum levels of vitamin B12 not always represent deficiency, and likewise, severe functional deficiency of the micronutrient has been documented in the presence of normal and even high levels of serum vitamin B12. This review discusses the usefulness and limitations of current biomarkers of B12 status in newborn screening, infant and adult diagnostics, the algorithms utilized to diagnose B12 deficiency and unusual findings of vitamin B12 status in various human disorders.
Vitamin B12 Deficiency
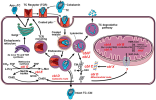
Vitamin B12 (B12 = Cbl, “cobalamin,” the chemical name) is an essential water-soluble micronutrient required by all cells in the body. Humans are unable to synthesize B12 and thus rely on dietary intakes and a complex intracellular route for vitamin processing and delivery to its target destinations (Figure 1) (Hannibal et al., 2009). Vitamin B12 deficiency due to malabsorption and inadequate intake is a public health issue worldwide. It is estimated that 15–20% of the elderly in the United States are B12 deficient (Allen, 2009). In Germany, about 10% of the male elderly population and 26% of the female elderly population present with insufficient levels of vitamin B12 (Hartmann, 2008; Grober et al., 2013). In India, ~75% of the population, i.e., over 650 million people, have B12 deficiency (Antony, 2001; Refsum et al., 2001), which can only be partly ascribed to a vegetarian diet in a substantial portion of the population.
Vitamin B12 deficiency is a multifactorial condition caused by insufficient intake (nutritional deficiency) as well as acquired or inherited defects that disrupt B12 absorption, processing and trafficking pathways (functional deficiency). Methylcobalamin (MeCbl) serves as a coenzyme for the biosynthesis of methionine from homocysteine catalyzed by the cytosolic enzyme methionine synthase (MS). This reaction regenerates tetrahydrofolate (THF) from N5-methyl-tetrahydrofolate (N5-CH3-THF), which is essential for the de novo biosynthesis of nucleic acids. Adenosylcobalamin (AdoCbl) is required for the conversion of methylmalonyl-CoA to succinyl-CoA catalyzed by mitochondrial methylmalonyl-CoA synthase (MCM), an anaplerotic reaction that furnishes increased demands for the Krebs cycle and heme biosynthesis precursor succinyl-CoA.
Insufficient supply of B12 and genetic defects impairing its cellular processing and trafficking lead to the accumulation of homocysteine (Hcy) and methylmalonic acid (MMA), which enter circulation and give rise to hyperhomocystinemia and methylmalonic acidemia.
[My note: What this basically means is that you need B12 to be functioning inside the cells to recycle homocysteine and methylmalonic acid. Hence, if either of these markers test as high, then this demonstrates a functional deficiency. In this context, you can have perfectly normal B12 levels in serum, but it is not being used inside the cells properly. The end result is a deficiency on the intracellular level]
Vitamin B12 deficiency is frequently under-diagnosed in pregnancy and in infants from mothers having insufficient levels of the micronutrient (Wheeler, 2008; Sarafoglou et al., 2011). Ensuring sufficient intake of vitamin B12 during pre-conception, pregnancy, and post-partum is strongly recommended (Bjørke Monsen et al., 2001; Rasmussen et al., 2001; Bjørke-Monsen et al., 2008; Hinton et al., 2010; Dayaldasani et al., 2014). Other populations at risk of developing vitamin B12 deficiency include the elderly, vegetarians and vegans, recipients of bariatric surgery (Majumder et al., 2013; Kwon et al., 2014) as well as those suffering from gastrointestinal diseases featuring ileal resections >20 cm (Battat et al., 2014). Certain medications such as metformin (Greibe et al., 2013b; Aroda et al., 2016) and proton-pump inhibitors (Howden, 2000; Wilhelm et al., 2013) may also transiently induce a status of cobalamin deficiency, which may be reversible upon completion of treatment and/or with oral vitamin B12 supplementation.
Herein, we discuss three aspects of the assessment of B12 status: (1) The utility of metabolites used as biomarkers of vitamin B12 deficiency in neonates and adults, (2) The algorithms employed to predict subclinical and clinical B12 deficiency, and (3) Major challenges and diagnosis of vitamin B12 deficiency in special populations.
Serum Biomarkers of Vitamin B12 Deficiency: Strengths and Limitations
Total Serum Vitamin B12
The most direct assessment and perhaps preferred first-assay to determine vitamin B12 status is the measurement of total serum vitamin B12. This assay is widely available in clinical chemistry laboratories. Ranges for normal (>250 pmol/L), low (150–249 pmol/L), and acute deficiency (<149 pmol/L) vitamin B12 have been defined and are used in most clinical chemistry laboratories worldwide (Clarke et al., 2003; Selhub et al., 2008; Mirkazemi et al., 2012). One limitation of this biomarker is that it assesses total circulating vitamin B12, of which ~80% is bound to haptocorrin, and therefore, not bioavailable for cellular uptake. Another limitation of this assay lies in its unreliability to reflect cellular vitamin B12 status. Results from studies assessing serum and cellular vitamin B12 have shown that the levels of serum B12 do not always represent cellular B12 status (Carmel, 2000; Solomon, 2005; Devalia et al., 2014; Lysne et al., 2016). In particular, patients with inborn errors of vitamin B12 metabolism can present with normal or low serum values of the vitamin, while being deficient at the cellular level. Furthermore, functional vitamin B12 deficiency due to oxidative stress has been identified in elders exhibiting normal values of serum vitamin B12 (Solomon, 2015). Functional deficiency of vitamin B12 was corrected upon supplementation with cyanocobalamin (CNCbl), as judged by reduction in the serum levels of tHcy and MMA (Solomon, 2015). Thus, total serum B12 is not a reliable biomarker of vitamin B12 status when used alone. Nonetheless, this marker should not be considered obsolete as a number of studies show that total serum vitamin B12 may be helpful to predict prognosis and status of diseases featuring abnormally high vitamin B12 levels in serum (>650 pmol/L), such as cancer (Arendt et al., 2016) and autoimmune lymphoproliferative syndrome (ALPS) (Bowen et al., 2012).
Homocysteine
Homocysteine is a metabolite of one-carbon metabolism that is remethylated by MeCbl-dependent MS or betaine-homocysteine methyltransferase as part of the methionine cycle (Finkelstein and Martin, 1984) and degraded by cystathionine β-synthase (CBS) in the transsulfuration pathway (Figure 2). Conversion of Hcy to Met by MS depends on the availability of both vitamin B12 and folate (as N5-CH3-THF), and therefore, nutritional deficiencies in either one of these micronutrients lead to the accumulation of Hcy in serum and urine. Likewise, inborn errors of metabolism that impair the upstream processing and trafficking of B12 or folate lead to elevation of this metabolite, a condition collectively known as hyperhomocystinemia. The normal range of total plasma Hcy (tHcy) in human plasma is 5–15 μmol/L (Ueland et al., 1993) and values >13 μmol/L may be considered elevated in adults (Jacques et al., 1999). Homocysteine levels are always higher in serum compared to plasma due to the release of Hcy bound to cellular components (Jacobsen et al., 1994). Hence, plasma and not serum should be used to determine the levels of tHcy. Although gender and age reference intervals have been established in some studies (Jacobsen et al., 1994; Rasmussen et al., 1996; van Beynum et al., 2005), they are usually ignored in the reporting of tHcy levels. Because of the dual biochemical origin of elevated Hcy, this biomarker is of limited value to assess vitamin B12 status as a stand-alone measurement. This is also true for the newborn screening of inborn errors of vitamin B12 metabolism found in the cblD, cblF, and cblJ (Huemer et al., 2015) disorders.
Figure 2. Pathways for Hcy and MMA metabolism in humans. (A) Homocysteine is a branch-point metabolite at the intersection of either the remethylation or the transsulfuration pathways. Thus, Hcy homeostasis relies on three different biochemical reactions [MS, cystathionine β-synthase (CBS) and S-adenosylhomocysteine hydrolase (SAHH)], two of which (CBS and SAHH) are independent of vitamin B12. In addition to nutritional deficiency of vitamin B12, elevation of Hcy in plasma may arise from reduced function of CBS and MTHFR, as well as nutritional deficiencies of folate. (B) MMA is produced during catabolism of odd-chain fatty acids and amino acids in the mitochondrion. Propionyl-CoA is the precursor of MMA in a reaction catalyzed by propionyl-CoA carboxylase (PCC). Inborn errors of PCC lead to propionic acidemia. Likewise, mutations in AdoCbl-dependent MCM lead to a buildup of MMA-CoA and inhibition of PCC that manifests as increased propionyl-CoA and so of propionic acid the circulation. Propionylcarnitine can also be transported out of the cell to reach systemic circulation. Propionylcarnitine is a first-line test in newborn screening.
Methylmalonic Acid
MMA increases upon inactivation of AdoCbl-dependent MCM in the mitochondrion. Nutritional and functional deficiencies of vitamin B12 result in the inactivation of MCM leading to buildup of its substrate methylmalonyl-CoA, which enters circulation as free MMA. The reaction catalyzed by MCM (Figure 2) is not affected by other vitamins of one-carbon metabolism, and therefore, MMA is considered a more specific marker of vitamin B12 deficiency (Clarke et al., 2003). Serum values of MMA, ranging from >260 to 350 nmol/L indicate elevation of this metabolite (Clarke et al., 2003). Nonetheless, there are few pathologies such as renal insufficiency that lead to an increase in MMA (Iqbal et al., 2013). For example, one study showed that 15–30% of individuals with high vitamin B12 concentrations in serum also had elevated MMA concentrations, which may reflect renal dysfunction instead of authentic vitamin B12 deficiency (Clarke et al., 2003). Thus, the utility of this marker should be considered carefully in older patients and patients with suspected or established renal disease. Assessment of a second marker of vitamin B12 status, such as holo-transcobalamin (holo-TC) (Iqbal et al., 2013) should be considered. Another study showed that the clearance of both Hcy and MMA may be compromised in patients with reduced kidney function (Lewerin et al., 2007).
Total Serum Holo-Transcobalamin
Dietary B12 is transported in the digestive system via the use of three protein transporters that bind the micronutrient in a sequential fashion, following the order haptocorrin (HC), intrinsic factor (IF), and transcobalamin (TC) (Fedosov et al., 2007; Fedosov, 2012). After absorption in the intestine, vitamin B12 bound to TC (holo-TC) reaches circulation and it is distributed to every cell in the body. Cells take up holo-TC via receptor-mediated endocytosis, aided by the transcobalamin receptor (TCblR; CD320) (Quadros et al., 2009). Because the only fraction of dietary vitamin B12 that is bioavailable for systemic distribution is in the form of holo-TC (Valente et al., 2011), the level of holo-TC in serum has been successfully utilized as a marker of bioactive vitamin B12 (Nexo et al., 2000, 2002; Valente et al., 2011; Yetley et al., 2011). Holo-TC represents 6–20% of the total vitamin B12 present in serum (Nexo et al., 2000, 2002; Valente et al., 2011; Yetley et al., 2011). This marker is more accurate in assessing the biologically active fraction of vitamin B12 in circulation than serum B12 itself, and its level correlates well with the concentration of vitamin B12 in erythrocytes (Valente et al., 2011). The diagnostic value of holo-TC has proven superior to Hcy and MMA for the assessment of vitamin B12 status in the elderly (Valente et al., 2011). The normal range of holo-TC in healthy subjects is 20–125 pmol/L (Valente et al., 2011). Additional research is needed to elucidate the mechanisms that control holo-TC homeostasis in the normal population and in pathologies that alter vitamin B12 transport and utilization. For example, abnormally low levels of holo-TC have been documented in patients receiving chemotherapy, macrocytosis and in individuals carrying the TC polymporphism 67A>G, without vitamin B12 deficiency (Vu et al., 1993; Wickramasinghe and Ratnayaka, 1996; Riedel et al., 2005, 2011). Insufficient sensitivity (44%) of holo-TC as a marker of vitamin B12 status was noted in a cohort of 218 institutionalized elderly patients (Palacios et al., 2013). At present time, it is unknown whether and how holo-TC levels vary in patients harboring inborn errors affecting intracellular vitamin B12 metabolism (cblA-cblJ). Thus, the diagnostic value of holo-TC as a first line test awaits further investigation.




 :
:
